
Human brains are wired to respond well to immediate threats from other humans or animals.
We are not good at responding to gradual, long-term threats that we cannot see.
The threat to your health from the coronavirus COVID-19 is real, especially if you are elderly or have a compromised immune system. The threat that your lifestyle will be significantly disrupted by COVID-19 is imminent. Work, school, vacations, concerts, sporting events and even our upcoming elections are at risk. The financial markets are showing signs of the expected impact on businesses.
COVID-19 is not a China-only problem anymore. It has gone global with recent cases here in the United States in California, Oregon, Washington, Rhode Island, Florida and New York. The outbreak in Northern Italy during the 100,000 person international festival has accelerated the spread around the world. John Hopkins University has set up this useful tracking tool for the virus. As of March 3, 2020, Italy, Iran and South Korea had over 1000 cases each. Epidemic clusters were spreading around the world with over 93,000 cases and 3000 deaths. 51,000 people have recovered. As of March 17, 2020, the number of world-wide cases has doubled from ~90,000 to ~180,000 and COVID-19 is officially a pandemic. The number of countries with over 1,000 cases has gone from three to sixteen. The number of recovered patients has gone from 51,000 to 78,000. The number of deaths has risen from 3,000 to 7,000. This isn’t good news.
Prior pandemics, such as the 2009-2010 H1N1 swine-flu also spread worldwide. The swine-flu caused 18,500 laboratory-confirmed deaths with an estimated 151,700 to 575,400 total deaths. In August 2010, the World Health Organization (WHO) declared the H1N1 influenza pandemic over, saying worldwide flu activity had returned to typical seasonal patterns. H1N1 had become a mild to moderate flu for most people. Unfortunately, COVID-19 does not appear to be a seasonal epidemic. It has spread in Hong Kong with Fahrenheit temperatures in the 70s.
WHO explains that people can catch COVID-19 from others who have the virus. The disease can spread from person to person through small droplets from the nose or mouth which are spread when a person with COVID-19 coughs or exhales. These droplets land on objects and surfaces around the person. Other people then catch COVID-19 by touching these objects or surfaces, then touching their eyes, nose or mouth. People can also catch COVID-19 if they breathe in droplets from a person with COVID-19 who coughs out or exhales droplets. This is why it is important to stay more than 2 meters (6 feet) away from a person who is sick and more than 30 feet away from anyone who is coughing or sneezing.
Hand washing, maintaining distance from people who are coughing, and not touching one’s face with unwashed hands are measures recommended to prevent the disease. As with colds and flu symptoms, it is recommended to cover one’s nose and mouth with a tissue or a bent elbow when coughing. Even if you aren’t concerned about COVID-19, these are good practices as the seasonal flu has killed at least 18,000 people in the United States so far this season, according to the CDC.
It is also important to boost your immune system by:
- Getting enough sleep
- Eating fruits and vegetables
- Not smoking
- Minimizing alcohol consumption
- Avoiding sugar
- Exercising, but not to exhaustion
- Minimizing stress
With COVID-19, time from exposure to onset of symptoms is generally between 0 and 14 days, with an average of 5-6 days. People may have few symptoms or develop fever, cough, and shortness of breath. Cases can progress to pneumonia and multi-organ failure. To prevent transmission, the CDC recommends that infected individuals stay at home except to get medical care; call ahead before visiting a healthcare provider; wear a facemask (especially in public); cover coughs and sneezes with a tissue; regularly wash hands with soap and water; and avoid sharing personal household items.
According to WHO, based on analysis of 44,000 cases of COVID-19 in Hubei province, around 80% of people have a mild form of the disease, 14% developed more severe disease such as pneumonia, 5% have critical disease, and 2% of cases are fatal. Here are CDC’s fatality rates by age from China as of February 26, 2020.
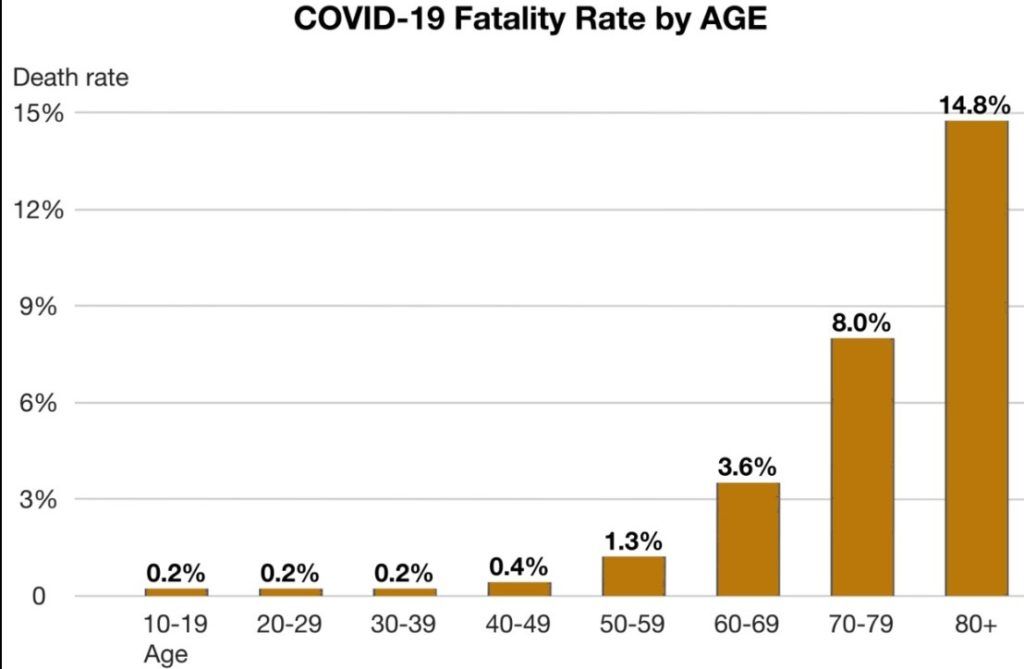
Among those who died, many had preexisting conditions, including hypertension, diabetes, or cardiovascular disease, and the median time from initial symptoms to death was 14 days (range 6–41 days). Males had a death rate of 2.8% while females had a death rate of 1.7%. In those under the age of 50 the risk of death is less than 0.5% while in those over the age of 70 it is more than 8%. No deaths had occurred under the age of 10.
It is important to understand why COVID-19 can be fatal and what options are emerging for treatment of severe cases.
Although most people have mild symptoms, those with pre-existing conditions as well as older adults are most at risk for a severe and possibly fatal illness. There is emerging evidence that the primary cause of death from COVID-19 occurs because of a bad immune system response. This malfunction creates a cytokine storm from over-production of IL-6, a powerful pro-inflammatory. This leads to Adult Respiratory Distress Syndrome (ARDS) which occurs when fluid builds up in the tiny, elastic air sacs (alveoli) in your lungs. The fluid keeps your lungs from filling with enough air, which means less oxygen reaches your bloodstream. This deprives your organs of the oxygen they need to function. The risk of death increases with age and severity of illness. Of the people who do survive ARDS, some recover completely while others experience lasting damage to their lungs.
In times of crisis, it is good to have hope and even better to have talented scientists.
Fortunately, 12 companies have created anti-IL-6 drugs to stop cytokine storms. The first approved drugs were created for treating lung cancer and rheumatoid arthritis.
In a small single-arm, 20-patient Chinese trial in severely ill COVID-19 patients, most patients experienced rapidly reduced fevers and 75% (15 out of 20) reduced their need for supplemental oxygen within days of receiving an IL-6 receptor antibody from Roche called ACTEMRA (tocilizumab). Based on these results, China updated its COVID-19 treatment guidelines and approved the use of tocilizumab to treat patients with severe or critical disease. The timing of treatment is important. It must be provided early enough to avoid lung damage.
Of note and rather ironically, anti-IL-6 drugs have side-effects including the risk of serious infections. 🙁
The twelve companies are working actively with governing bodies to provide assistance for severely ill-patients through rapidly created COVID-19 clinical trials. Beginning on March 16th, Regeneron’s Kezvara is being provided to 400 patients in 16 states. The first segment of the trial will study Kevzara’s impact on fever and patients’ need for supplemental oxygen while a second segment will focus on longer-term outcomes, including preventing death and cutting the need for supportive care such as mechanical ventilation, supplemental oxygen and/or hospitalization. Regeneron has the capability to rapidly produce Kezvara. Regeneron is also working on a cocktail of virus-neutralizing antibodies for an even more effective treatment.
There is also hope on the vaccine side as Moderna, in collaboration with the National Institute of Health, initiated test injections of COVID-19 vaccine in Seattle also starting on March 16th. Moderna also has rapid large-scale manufacturing capability. lnovio Pharmaceuticals’ vaccine is expected to be ready for testing next month. Other companies continue to make progress as well.
For those interested, here are some companies working on drug treatments and vaccines:
- Altimmune (ALT)
- Clover Biopharmaceuticals (private)
- CureVac (private)
- CytoDyn (CYDY)
- Gilead Sciences (GILD) initiated two phase 3 trials for its drug remdesivir which was active against the SARS and MERS viruses in preclinical tests. COVID-19 is genetically similar.
- GlaxoSmithKline (GSK)
- Inovio Pharmaceuticals (INO)
- Johnson & Johnson (JNJ)
- Moderna (MRNA)
- Novavax (NVAX)
- Regeneron Pharmaceuticals (REGN) is working with the National Institute of Allergy and Infectious Diseases (NIAID).
- Sanofi (SNY)
- Vaxart (VXRT)
- Zhejiang Hisun Pharmaceuticals (China) is testing COVID-19 patients with favilavir, a drug approved for influenza.
It’s difficult to predict which companies will be successful.
Since it is likely that COVID-19 will spread to your community, see the recommendations from the Center for Disease Control.
Additionally, Keep Health recommends stockpiling enough food and household supplies in case you need to shelter in place or wait out supply-chain disruptions. Those most at-risk from COVID-19 with daily jobs or activities in public places must think seriously about giving those up. To slow the outbreak, European countries are closing public places such as the Louvre and soccer stadiums. Although catching COVID-19 may be inevitable, delaying that occurrence until modern medicine is better prepared seems wise.
The COVID-19 outbreak is unlikely to be a temporary problem. It will take at least a year to develop and test a vaccine, if that is even possible as COVID-19 has the potential to develop numerous strains. Over a dozen life sciences companies are actively working on a solution. These companies will require much more time to develop sufficient quantities of the vaccine along with methods to transport and distribute it globally. In the meantime, our health care systems are not built to handle 20% of the population becoming very ill. The COVID-19 outbreak has the potential to disrupt our lives significantly for several years. We hope this does not happen.
In terms of other things to consider in planning for COVID-19, those with investment and/or retirement accounts need to be careful. It is not hard to imagine how travel restrictions, quarantines and large numbers of contagious sick employees will be disruptive for businesses.
This concludes today’s article from Keep.Health. As your reward for reading, we recommend you order some healthy and tasty seaweed salad from Maine to boost your immune system and support local farmers. It is surprisingly good and much better than the dyed seaweed salads from Asia.
Sources:
Information in this article was sourced from the COVID-19 sites on:
- Wikipedia
- World Health Organization
- CNN
- Center for Disease Control
- Center for Infectious Disease Research and Policy
and
